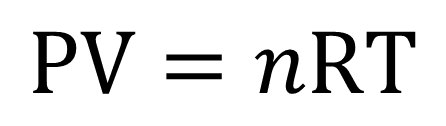WHat is the Ideal Gas LAw?
The Ideal Gas Law is a gas law that describes the relationship among volume, pressure, temperature, and amount in moles of an ideal gas: PV=nRT
It was derived from the combined gas law and Avogadro’s law, meaning that when rearranged, for a given variable, it forms the equations of Boyles law and Charles law.
It was derived from the combined gas law and Avogadro’s law, meaning that when rearranged, for a given variable, it forms the equations of Boyles law and Charles law.
Uses of Ideal Gas Law:
The ideal gas law is useful for predicting/calculation the properties of a gas under ordinary conditions, with accuracy within 5%. (When gases are behaving “ideally”.) It serves as a model for gas behaviours.
Ideal Gas law does not apply at higher than critical temperature/low pressures.
The ideal gas law is a statement that concerns how gas reacts in response to changes in temperature, pressure, volume.
Ideal Gas law does not apply at higher than critical temperature/low pressures.
The ideal gas law is a statement that concerns how gas reacts in response to changes in temperature, pressure, volume.
IT Describes:The volume (V) that is occupied by n number of moles a gas has at a pressure (P) at temperature (T) in Kelvin.
“R” is known as the gas constant, which is the ideal gas law or equation of state. The ideal gas law shows that :
"R" - The Universal Gas constant“R” is known as the gas constant, which is the ideal gas law or equation of state.
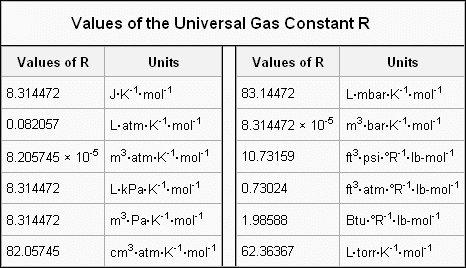
Repeated experiments show that at standard temperature (273 K) and pressure (1 atm ), one mole (n = 1) of gas occupies 22.4 L volume. Using this experimental value, you can evaluate the gas constant R, R = PV/nT = 1 atm ×22.414 L/(1 mol × 273.15 K) = 0.082 057 L•atm•K⁻¹mol⁻¹ Depending on the different units used for Pressure and Volume, the constant of R will be different. |
Different values of R based on various Pressure/Volume units
|
Guidelines for using the ideal gas law:
|
1. Always convert temperatures to Kelvin units (K). |
2. Always convert the mass to moles. |
|
3. Always convert the volume to litres (L).
|
4. Preferably, use kPa (kilopascals) for pressure. This way, the value of R will always be 8.314 kPa.L/mol.K
|