|
How it works:
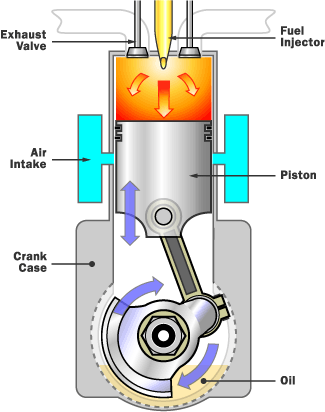
The internal combustion engine works on the principle of putting a small amount of high energy fuel into a small enclosed space, and then igniting it. This will result in an large amount of energy being produced in the form of expanding gas. There are several types of internal combustion engines, depending on the vehicle. Some examples are: diesel engines, gas turbine engines, etc. Most cars nowadays use a four-stoke combustion cycle to convert gasoline into motion.
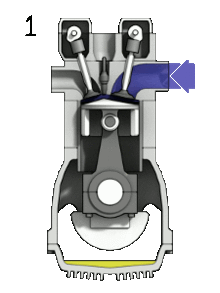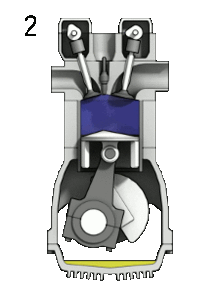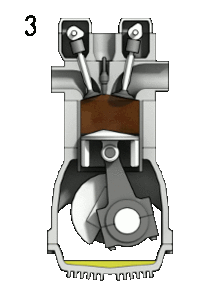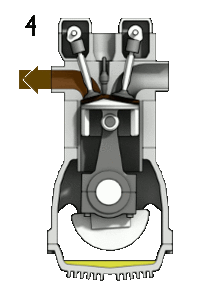
The image on the right describes the part of a four stoke engine:
The device called the piston is connected to the crankshaft by the connecting rod. The crankshaft rotates, and the engine goes through its cycle. |
Internal combustion engine (four stoke engine) and the gas laws

1. Intake stoke/ Induction
Firstly, the piston of the engine starts at the top, moving down to allow an intake of air and gasoline. This increases the volume of the combustion chamber. A small amount of gasoline is required for the cycle to work. The mixture of air and fuel is called the charge.
2. Compression
this is the second stoke of the engine, in which the piston moves upwards. This reduces the volume of the combustion chamber. The gas is being compressed. The piston works on the charge . As a result, the pressure, temperature and density increases .
Firstly, the piston of the engine starts at the top, moving down to allow an intake of air and gasoline. This increases the volume of the combustion chamber. A small amount of gasoline is required for the cycle to work. The mixture of air and fuel is called the charge.
2. Compression
this is the second stoke of the engine, in which the piston moves upwards. This reduces the volume of the combustion chamber. The gas is being compressed. The piston works on the charge . As a result, the pressure, temperature and density increases .
|
The volume of the compression chamber decreases. As a result, the pressure will increase. Because p x V must equal to nRT, where n remains constant, R and T will therefore also increase.
|
p↑V↓ = n R↑T↑
|
3. Combustion
When the piston reaches the top, a spark is emitted to ignited the gasoline, and it explodes. This drives the piston down once again.
Ideally an engine takes in air (Oxygen and Nitrogen) and fuel (hydrocarbons) and produces CO2, H2O. The N2 just passes straight through. The chemical equation is as follows:
2 C8H18 (g) + 25 O2 = 16 CO2 + 18H2O
4. Exhaust
Once the piston is at the bottom once again, the exhaust valve opens and the exhaust will leave the cylinder out of the tailpipe of the vehicle.
Other technologies related to the ideal gas law
Chemical and Petrochemical plants |
The Ideal Gas Law helps engineers discover and design chemical and petrochemical plants used in different industries, by exploring the different properties of gases through this law. This knowledge helps make the plants work efficiently.
|
Gas pipelinesTo transfer gases, natural gas companies rely on gas pipelines. Unlike oil, natural gas is delivered directly to homes and businesses through pipelines. To determine the capacity and pressure inside the gas pipes, engineers use the ideal gas law.
The natural gas liquids and the natural gases are separated from the contaminants. Once it’s processed, natural gas pipeline operators ship the natural gas through large transmission pipelines. Compressor stations are located along the pipeline route. These large compressors are used to move natural gas through the pipeline at around 40 km an hour. |